Verifying Natural Products: Is Carbon-14 Analysis Necessary?
 As natural products become more popular, so follows the increasing incentive for adulteration. There are many testing methods available to help authenticate natural products. However, owing to the complexity of many natural products and new adulteration methods, there is no single test that can guarantee 100% authenticity.
As natural products become more popular, so follows the increasing incentive for adulteration. There are many testing methods available to help authenticate natural products. However, owing to the complexity of many natural products and new adulteration methods, there is no single test that can guarantee 100% authenticity.
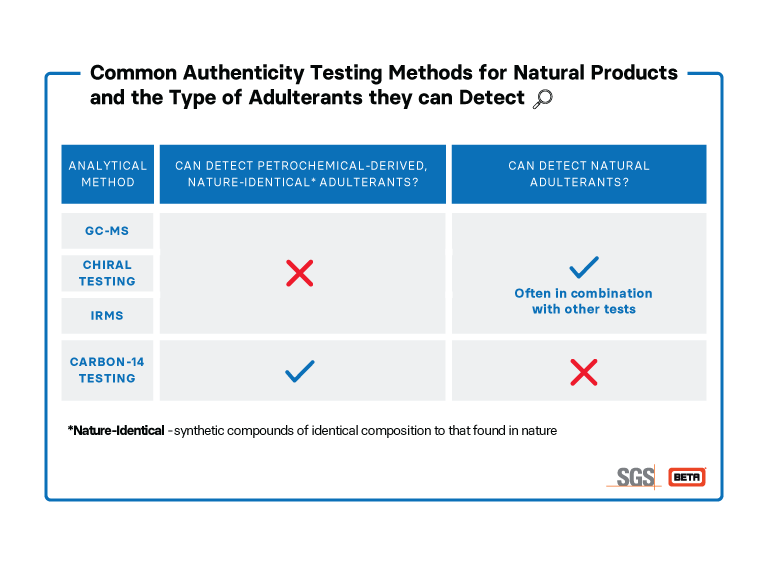
While there are several methods that can detect if a product contains adulterants from other natural sources, only Carbon-14 analysis can confirm if a material has synthetic, petroleum-sourced adulterants. Thus, to ensure that an ingredient doesn’t have both natural and petrochemical-derived adulterants, Carbon-14 analysis is a necessary tool for quality control.
Gas Chromatography – Mass Spectrometry (GC-MS)
GC-MS uses two different testing methods, namely gas chromatography and mass spectrometry. Gas chromatography can separate complex volatile mixtures into its various components. By sending the components through a mass spectrometer, the compounds are ionized and broken into smaller fragments which the spectrometer will record the masses and relative amounts of. By comparing the resulting spectrum with a reference database, the composition and authenticity of a sample can be assessed. As a sensitive technique, GC-MS is popular for the testing of many products, including essential oils and fragrances.
Chiral Testing
Chirality is a property of some molecules that relates to their 3D orientation in space. Chiral molecules (with one chiral center) exist in two configurations that are non-superimposable mirror images of each other, comparable to a left and right hand – they have the same components (or fingers) but cannot be superimposed.
In nature, it is often one “hand” that is found. The synthetic equivalents made in a lab are more often a racemic mixture (an even ratio between the left and right-handed forms of the compound). Chiral testing is able to distinguish between a racemic mixture and the presence of just one of the left or right-handed forms.
There are several methods that can examine the chirality of a compound. The simplest of these is optical rotation – the left-handed form will rotate light in one direction and the right-handed form will rotate light the same amount but in the opposite direction. A racemic mixture will not rotate light at all, as the rotations from the left and right handed forms cancel. By comparison with a reference value, the chiral purity can be determined.
Isotope Ratio Mass Spectrometry (IRMS)
Elements can exist in different isotopes – having the same number of protons but a different number of neutrons and consequently a different atomic mass. For example, the most naturally abundant isotope of carbon is carbon-12, containing 6 protons and 6 neutrons. However, carbon-13, with 6 protons and 7 neutrons, also exists though at a lower abundance. Measuring the ratio between different isotopes of a particular element can often reveal a lot about the source of the sample and is used widely from the authentication of honey to identifying sources of nitrate pollution.
Isotope ratios can be measured using an Isotope Ratio Mass Spectrometer which ionizes samples and sends them through a magnetic field. Molecules are deflected differently depending on their mass and are then picked up by a detector. By comparing with an authentic value for the sample, various isotope ratios can reveal the presence of any foreign materials that would skew the ratio.
Carbon-14 Analysis
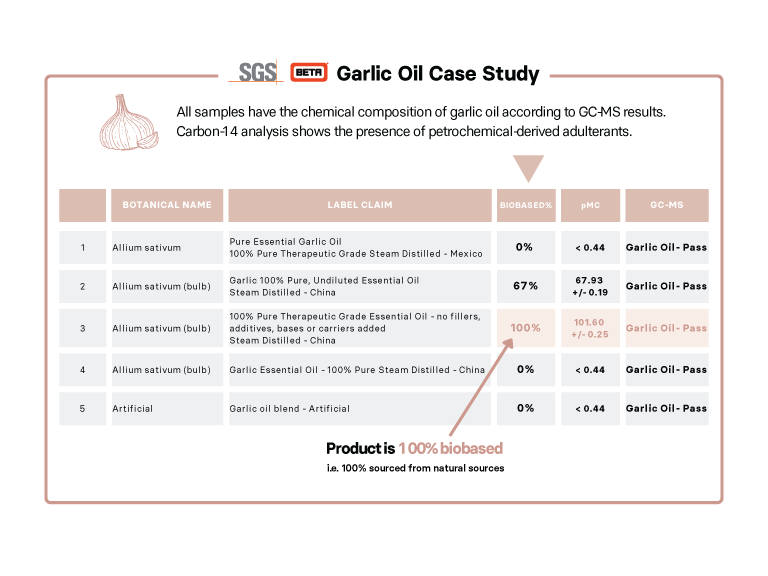
One of the methods of adulteration of natural products (of plant or animal origin) is using the often cheaper petrochemical-derived synthetic version. This may be identical in chemical composition to the natural source but adulteration can be detected using carbon-14 analysis. A weakly radioactive carbon isotope, carbon-14 decays over time according to the law of radioactive decay. As part of the carbon cycle, all living things have a known level of carbon-14, whereas petrochemical-derived compounds have been out of the carbon cycle for a sufficiently long time that they do not contain any carbon-14. Thus, by measuring the carbon-14 content of a product, its petrochemical-derived sources can be distinguished from plant or animal sources. Carbon-14 testing, however, cannot distinguish between different natural sources, for example between plant and animal-sourced material.
Carbon-14 is measured using an Accelerator Mass Spectrometer which works on similar principles to a standard mass spectrometer described above. The key difference is that it accelerates the ions to a much higher speed before they reach the detector and this allows the detection of very low abundance isotopes, for example separating and detecting carbon-14 relative to carbon-12 and carbon-13.
Disclaimer: This video is hosted in a third-party site and may contain advertising.
Excerpt from Beta Analytic’s presentation during the webinar “Stewarding Responsible Growth by Protecting the Integrity of the Curcumin Category”
Conclusion
The testing methods available today offer a great arsenal for those in the natural product industry to ensure the authenticity of their materials. This is of course not a fail-safe guarantee. By employing several testing methods that detect different issues, quality control is enhanced.
References:
G. Downey (ed.), 2016, Advances in Food Authenticity Testing, Amsterdam: Woodhead Publishing.
Zawirska-Wojtasiak, R, 2006, Chirality and the Nature of Food Authenticity of Aroma, Acta Scientiarum Polonorum Technologia Alimentaria, 5, pp. 21 – 36.
This entry was posted on Tuesday, April 10th, 2018 and is filed under Essential Oil Testing, Flavor & Fragrance, Flavors Carbon-14 Analysis, Natural Product Testing, Supplement Carbon-14 Testing .