The USDA BioPreferred Program
- The USDA BioPreferred® Program aims to aid consumers easily identify commercially available biobased products.
- Applicable to both domestic and imported products, the Program requires ASTM D6866 testing from a USDA-approved laboratory.
- ISO/IEC 17025:2017-accredited Beta Analytic has been approved by the USDA to provide ASTM D6866 testing for BioPreferred applicants.
Ready to send samples?
Please read the lab’s instructions for USDA Certified Biobased Product Label Applicants
 Note to all Applicants – Please make sure your Biobased Participant Agreement has been signed and accepted. If you obtain an analysis prior to acceptance, the USDA will not accept the result even if you have an application number. A tutorial on Label Application is found in the USDA BioPreferred Program website.
Note to all Applicants – Please make sure your Biobased Participant Agreement has been signed and accepted. If you obtain an analysis prior to acceptance, the USDA will not accept the result even if you have an application number. A tutorial on Label Application is found in the USDA BioPreferred Program website.
Are you sending Intermediates or Retail Products?
Intermediates – If you have listed your material on the USDA Certified Biobased application as an intermediate product, it is okay to sub-sample and deliver in any packaging appropriate for the material. Our laboratory will take a picture once we receive the sample. “Intermediate” is a raw material which is sold then modified or used in making a “retail” product which is then purchased by the consumer.
Retail Products – If you have listed your material on the USDA Certified Biobased application as a retail product, please send a sample in its retail packaging (usually the smallest available). “Retail” is defined as a product that is purchased directly by the consumer or is a packaging used “as-is” to contain a product purchased by the consumer. The laboratory is required to take a picture of this as third-party validation of what was analyzed (i.e. the result was obtained on material taken from the same container shown in the picture). If you would like to sub-sample a retail product before submitting it to the laboratory, please consult the USDA BioPreferred Program.
Beta Analytic can only accept samples that can be disposed of legally by pouring down the drain or throwing in the garbage in the state of Florida. Please consult the laboratory before sending your samples.
ISO/IEC 17025:2017-accredited Beta Analytic is not affiliated with the USDA BioPreferred Program. However, you are welcome to contact the lab on how to submit samples for testing as part of the Program.
BioPreferred Program’s Biobased Content Requirement
Created by the Farm Security and Rural Investment Act of 2002 (2002 Farm Bill) and expanded by the Agriculture Improvement Act of 2018 (2018 Farm Bill), the U.S. Department of Agriculture’s BioPreferred Program aims to increase consumption of biobased products in the United States.
Under this Program, successful applicants (manufacturers and vendors) can affix a distinctive USDA Certified Biobased Label on their goods and marketing materials as long as their products meet the BioPreferred Program’s requirements. The minimum biobased content has been established as part of the process of designating categories for federal purchasing preference by the USDA’s BioPreferred Program. Products that do not belong to the existing categories designated for federal purchasing are still eligible if they are at least 25% biobased. The minimum biobased content for all items are subject to change. The USDA BioPreferred Program intends to increase the minimum biobased content over time as biobased product technologies advance.
What are Biobased Products?
According to the USDA BioPreferred Program, biobased products are commercial or industrial products (other than food, feed or fuel) that are:
– composed, in whole or in significant part, of biological products, including renewable domestic agricultural materials, renewable chemicals, and forestry materials;
– an intermediate ingredient or feedstock.
Biobased products generally provide an alternative to conventional petroleum-derived products.
Click for USDA BioPreferred Program Product Categories & Minimum Biobased Content List
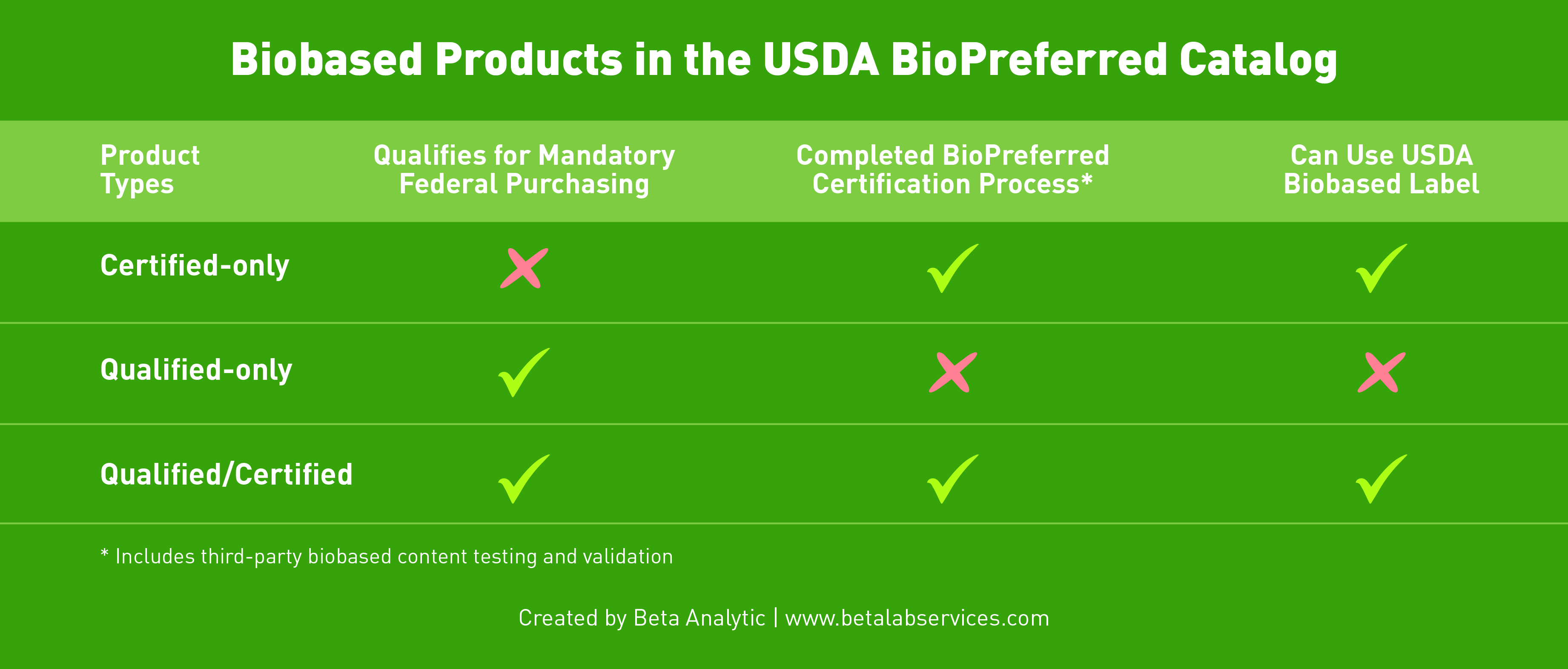 As of June 2022, there are three groups of biobased products in the BioPreferred Program: Certified-only (they do not fit into one of the 139 categories qualified for mandatory federal purchasing but have gone through the BioPreferred Program’s certification process); Qualified-only (they fall under one of the product categories but have not gone through the program’s certification process); and Qualified/Certified (they belong in one of the product categories AND have gone through the program’s certification process).
As of June 2022, there are three groups of biobased products in the BioPreferred Program: Certified-only (they do not fit into one of the 139 categories qualified for mandatory federal purchasing but have gone through the BioPreferred Program’s certification process); Qualified-only (they fall under one of the product categories but have not gone through the program’s certification process); and Qualified/Certified (they belong in one of the product categories AND have gone through the program’s certification process).
Video courtesy of the USDA • Disclaimer: This video is hosted in a third-party site and may contain advertising.
ASTM D6866 Biobased Testing Required
Other than product information, applicants are required to submit ASTM D6866 test results as evidence of the product’s biobased content. Applicants must also provide the USDA BioPreferred Program with the product’s brand names, contact information, and Web site URL (if available).
The Program intends to conduct occasional random ASTM D6866 testing on certified products to ensure compliance to minimum biobased content requirements.
MULTIPLE PRODUCTS – For products with similar biobased content but slightly different formulations, the USDA BioPreferred Program allows representative biobased content testing to suffice if the product formulation does not vary by more than 3 percent.
USDA Certified Biobased Label
 The USDA Certified Biobased Label consists of an artwork, the biobased content percentage, and either of these three text variations: USDA Certified Biobased Product, USDA Certified Biobased Product: Package, or USDA Certified Biobased Product and Package. The letters “FP” appear on the artwork of labels for products that qualify for mandatory federal purchasing.
The USDA Certified Biobased Label consists of an artwork, the biobased content percentage, and either of these three text variations: USDA Certified Biobased Product, USDA Certified Biobased Product: Package, or USDA Certified Biobased Product and Package. The letters “FP” appear on the artwork of labels for products that qualify for mandatory federal purchasing.
The USDA Certified Biobased Label is also applicable to products imported for sale in the U.S. as long as they meet the same criteria as U.S.-sourced biobased products. Even if they are considered to be biobased in their countries of origin, these imported products will carry the label only if they meet specific USDA BioPreferred Program criteria.
There is no specified deadline for application submissions. Applications to the USDA BioPreferred Program are evaluated on a first-come-first-serve basis. Companies can only use the label on the biobased product identified in their application when they receive the notice of certification from the USDA. Rejected applicants can submit revised applications for reconsideration.
Certification remains valid only if the certified product complies with the required minimum biobased content. If the USDA revises the required minimum levels, the certified product must meet the new requirement to continue using the label.
Source: Voluntary Labeling Program for Biobased Products (Final Rule), Federal Register Vol. 76 No. 13 Pages 3790-3813 Published January 20, 2011
Beta Analytic – The Leader in ASTM Method D6866 Testing
Beta Analytic joined the American Society for Testing Materials (ASTM) Subcommittee D20.96 as co-author of ASTM D6866 for biobased content analysis. The standard, adopted in July 2004, is an industrial application of radiocarbon dating. The current active version is ASTM D6866-22. The method is mentioned in the USDA BioPreferred Program as “ASTM Method D6866.”
The Center for Industrial Research and Service (CIRAS) at Iowa State University of Science and Technology, with the USDA at the helm, helped establish the required minimum biobased content for the BioPreferred Program. By June 2004, Beta Analytic has analyzed more than 200 biobased products for CIRAS and delivered the results within 1-3 weeks. ISO 17025-accredited Beta Analytic has proven to be a reliable service provider over the years.
Beta Analytic provides biobased content testing. The company is not affiliated with the USDA BioPreferred Program.
Page last updated: June 2022