Turmeric Case Study: Verifying “Natural” Claims by Carbon-14 and HPLC Analyses
Turmeric is a popular dietary supplement, herbal medicine and spice. This popularity has made turmeric and its curcuminoid derivatives vulnerable to economically motivated adulteration and false “natural” labeling. In collaboration with the Eurofins lab and the non-profit Global Curcumin Association, ISO 17025-accredited Beta Analytic recently published a case study using an orthogonal testing strategy to verify “all-natural” label claims of dietary supplements containing turmeric ingredients.
Potential Strategy for Turmeric Products Quality Control
Fourteen (14) samples of turmeric supplements sold in the US were selected randomly for this case study. All 14 products claimed to be “all-natural sourced.” To verify these claims, two analytical methods were used – High-Performance Liquid Chromatography (HPLC) and Carbon-14 testing by Accelerator Mass Spectrometry (AMS).
According to the study, a high percentage of curcumin-to-curcuminoids value was used as an indicator to imply the presence of synthetic curcumin. However, using the HPLC method alone does not provide direct evidence of the curcuminoids’ natural origin. The HPLC method is often used in the dietary supplements industry to accurately identify and quantify the chemical constituents in a finished product or raw material.
Carbon-14 analysis enhances quality control of dietary supplements by determining the percentage of material derived from biomass versus petrochemical-derived synthetic sources. However, the method cannot be used to verify potency claims nor determine which component of a product contains Carbon-14.
Analysis of results of both methods showed a significant correlation between the percentage of curcumin-to-curcuminoids and % biobased content, which indicated that synthetic curcumin was greatly attributed to determined synthetic ingredients.
Research proponents concluded that a strategy based on Carbon-14 and HPLC analyses has the potential to be used for routine quality control/assurance of turmeric products, with Carbon-14 analysis as the primary tool to determine the presence of synthetic ingredients and HPLC testing as secondary confirmation to indicate the spike of synthetic curcumin.
Disclaimer: This video is hosted in a third-party site and may contain advertising.
Turmeric Case Study Results
Nine out of the fourteen (9 out of 14) samples were entirely derived from biomass (i.e. 100% naturally sourced) based on Carbon-14 analysis results. The remaining five samples were a mixture of natural and fossil sources with results ranging between 83% and 91% of biobased content, which reveals false labelling claims.
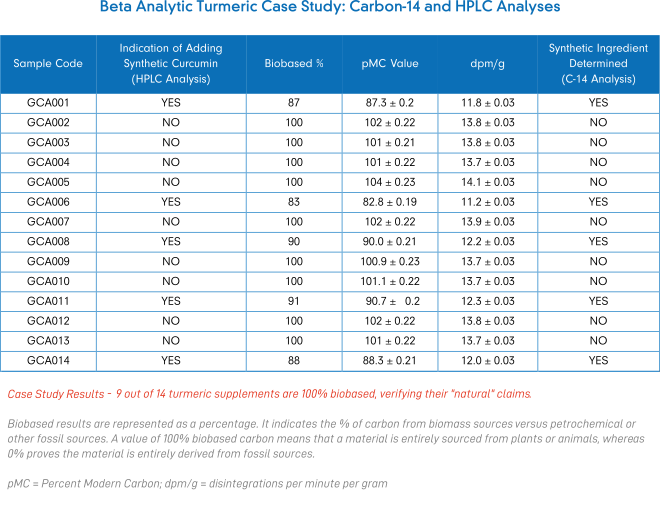
Based on the results of both analytical methods, only four samples (4 out of 14) proved to be accurate in relation to their potency and “natural” claims as promoted on their packaging labels.
The HPLC potency testing was done by Eurofins Botanical Testing using the USP Curcuminoids monograph methodology. Beta Analytic used the ISO 16620-2 analytical standard to measure the percentage of biomass-derived versus petroleum-sourced ingredients of the 14 samples.
For more information, please read the article “Analytical strategies to determine the labelling accuracy and economically-motivated adulteration of “natural” dietary supplements in the marketplace: Turmeric case study” published in Food Chemistry journal Volume 30 (https://doi.org/10.1016/j.foodchem.2021.131007).
Is Carbon-14 Analysis Necessary?
You might also be interested in this article – Verifying Natural Products: Is Carbon-14 Analysis Necessary?
Beta Analytic compared the different analytical methods used to test natural products: Gas Chromatography – Mass Spectrometry (GC-MS), Chiral Testing, Isotope Ratio Mass Spectrometry (IRMS), and Carbon-14 Testing.


Join our mailing list to receive news of upcoming webinars by Beta and its subsidiaries as well as other industry updates.
This entry was posted on Thursday, September 30th, 2021 and is filed under Natural Product Testing, SGS Beta Case Studies, SGS Beta Updates, Supplement Carbon-14 Testing .